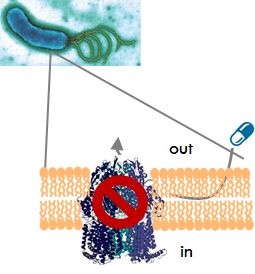
The main cause of multidrug resistance is the presence of drug efflux pumps, which remove antibiotics from the bacterial cell thereby lowering the antibiotic concentration inside the cells to sub-toxic levels. Drug efflux pumps recognise and expel a wide range of structurally unrelated compounds such as different classes of antibiotics, antiseptics, disinfectants and other biocides. The clinical consequence of this substrate promiscuity is multidrug resistance. Efflux pumps also play a role in the formation of bacterial biofilms and in pathogenesis. Hence, efflux pump inhibitors will (a) synergise with currently used antibiotics, (b) restore the efficacy of antibiotics to which resistance has arisen, (c) reduce the rate of emergence of drug-resistant pathogens, and (d) act as anti-biofilm and anti-virulence agents.
We are using our expertise on these efflux pumps to develop inhibitors that are reversing resistance and restoring antibiotic activity in some of the most critically dangerous and drug resistant pathogens.

Publications related to this project
- Wang Y, Alenzy R, Gu X, Polyak SW, Zhang P, Sykes MJ, Zhang N Venter H, Ma S (2021) Design and structural optimization of novel 2H-benzo[h]chromene derivatives that target AcrB and reverse bacterial multidrug resistance. Eur J Med Chem. 213:113049. doi: 10.1016/j.ejmech.2020.113049
- Gu X, Pisoni LA, Wang Y, Song D, Sykes MJ, Qin Y, Semple SJ, Polyak SW, Venter H, Ma S (2020) Design and synthesis of novel 4-substituted quinazoline-2-carboxamide derivatives targeting AcrB to reverse bacterial multidrug resistance. Bioorg Med Chem. 105: 104394. https://doi.org/10.1016/j.bioorg.2020.104394
- Wang Y, Alenzy R, Song D, Liu X, Teng Y, Mowla R, Ma Y, Polyak SW, Venter H*, Ma S, (2020) Structural optimization of natural product nordihydroguaretic acid to discover novel analogues as AcrB inhibitors. Eur J Med Chem, 186: 111910, doi.org/10.1016/j.ejmech.2019.111910
- Venter H* (2019) Reversing Resistance to Counter Antimicrobial Resistance in the World Health Organisation’s Critical Priority of Most Dangerous Pathogens. Biosci Rep, 39 (4). doi: 10.1042/BSR20180474.
- Jin C, Alenazy R, Wang Y, Mowla R, Qin Y, Tan JQE, Modi ND, Gu X, Polyak SW, Venter H*, Ma S (2019) Design, synthesis and evaluation of a series of 5-methoxy-2,3-naphthalimide derivatives as AcrB inhibitors for the reversal of bacterial resistance. Bioorg Med Chem Lett. 29(7):882-889. doi: 10.1016/j.bmcl.2019.02.003.
- Wang Y, Mowla R, Ji S, Guo L, De Barros Lopes MA, Jin C, Song D, Ma S, Venter H* (2018) Design, synthesis and biological activity evaluation of novel 4-subtituted 2-naphthamide derivatives as AcrB inhibitors. Eur J Med Chem, 143:699-709.
- Mowla R, Wang Y, Ma S, Venter H* (2018) Kinetic analysis of the inhibition of the drug efflux protein AcrB using surface plasmon resonance. BBA – Biomembranes, 1860:878-886.
- Arzanlou M, Chai WC, Venter H* (2017) Intrinsic, Adaptive and Acquired Antimicrobial Resistance in Gram-negative Bacteria. Essays in Biochem. 61:49-59.
- Wang Y, Mowla R, Guo L, Ogunniyi AD, Rahman T, De Barros Lopes MA, Ma S & Venter H* (2017) Evaluation of a series of 2-napthamide derivatives as inhibitors of the drug efflux pump AcrB for the reversal of antimicrobial resistance. Bioorg Med Chem Lett. 27:733-739.
- Wang Y, Venter H, Ma S (2016) Efflux Pump Inhibitors: A Novel Approach to Combat efflux-Mediated Drug Resistance in Bacteria. Curr Drug Targets. 17:702-19.
- Du D, Wang Z, James NR, Voss, JE, Klimont E, Ohene-Agyei T, Venter H, Chiu W, Luisi BF (2014) Structure of the AcrAB–TolC multidrug efflux pump. Nature. 509, 512-515.
- Ohene-Agyei T, Mowla R, Rahman T, Venter H* (2014) Phytochemicals increase the antibacterial activity of antibiotics by acting on a drug efflux pump. MicrobiolOpen, 3:885-896
- Du D, Venter H, Pos KM and Luisi BF (2013) The machinery and mechanism of multidrug efflux in Gram-Negative bacteria. In Microbial Efflux Pumps: Current Research, Eds. Yu EW, Zhang Q and Melissa H Brown MH, Caister Academic Press. Chapter 3, p 35–48.
- Ohene-agyei T, Lea J and Venter H* (2012) Mutations in MexB that affect the efflux of antibiotics with cytoplasmic targets. FEMS Microb Lett. 333, 20-27.
- Welch A, Chidebere U Awah, Shiheng J, van Veen HW and Venter H* (2010) Promiscuous partnering and independent activity of MexB, the multidrug transporter protein from Pseudomonas aeruginosa. Biochem J. 430, 355-364.
- Barrera NP, Isaacson SC, Zhou M, Bavro VN, Di Bartolo N, Welch A, Seeger MA, Schaedler TA, Nunez Miguel R, Korkhov VM, van Veen HW, Venter H, Walmsley AR, Booth PJ, Tate CG, and Robinson CV (2009). Mass spectrometry of integral membrane complexes reveals their subunit stoichiometry and interactions. Nature Methods. 6, 585-587.
You must be logged in to post a comment.